
Estimated reading time: 10 minutesI am delighted to introduce and share this guest blog from the Brain Inflammation Collaborative.
Matthew Menendez, PhD, has joined us to share his particular interest in complex chronic diseases. He has expertise in neuroinflammation and how this can be associated with conditions like Long Covid, myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), and autoimmune disorders.
This is really an area of emerging research, which I believe deserves more attention for our community, as so many of us have experience with these, along with hypermobility. And as we learn below, there may be a connection.
I am excited to explore this broader picture of potential inflammatory mechanisms linking hypermobility, Ehlers-Danlos Syndrome (EDS), mast cell activation syndrome (MCAS), post-viral illnesses, and brain inflammation.
In this blog, the role of chronic inflammation in the frequent overlap of symptoms will be examined. I am very grateful to the Brain Inflammation Collaborative for sharing their knowledge with us for this blog.
Key Takeaways
If you have hypermobility alongside conditions like ME/CFS, Long COVID, POTS, or MCAS, there may be a biological reason they so often appear together. Research shows joint hypermobility is found in 30–57% of people with these conditions — far above the 10–20% seen in the general population. Chronic inflammation may actually be driving connective tissue breakdown, not just accompanying it. This blog explores the emerging science behind these connections.
Key Terms & Acronyms
Why ME/CFS, Long COVID, POTS, MCAS, and EDS Don’t Fit the Standard Medical Model
Many medical conditions follow a straightforward clinical framework: one clear biological driver leads to a predictable set of symptoms, which doctors can often treat with FDA-approved drugs designed specifically for that condition.
Others are more complex. These complex chronic conditions (sometimes called complex or multisystem illnesses), such as ME/CFS, Long COVID, Ehlers-Danlos syndrome, POTS, and MCAS, can involve different biological drivers that cause a set of overlapping symptoms and comorbidities, leading to overlapping treatment responses from off-label drugs because, after all, there are no cures.
Here, we will examine the role of chronic inflammation in the frequent overlap of symptoms and comorbidities, including joint hypermobility (not explained by genetic mutations in connective tissue genes). But first, let’s define these complex chronic inflammatory diseases.
Complex Chronic Inflammatory Conditions Defined
Complex chronic conditions are long-lasting health issues (typically lasting at least 6–12 months) that affect multiple organ systems or severely impact a single system, leading to significant morbidity, functional limitations, and the need for ongoing, coordinated, multidisciplinary care.
These conditions are often further categorized by their primary drivers or features, including:
- Infection-associated chronic conditions (IACCs), triggered by acute infections and leading to persistent symptoms—examples include Long COVID, ME/CFS, and PTLDS (post-treatment Lyme disease syndrome).
- Dysautonomic conditions, involving autonomic nervous system dysfunction—such as POTS
- Immune-mediated or overactive immune response conditions, like MCAS

Overlapping Symptoms and Comorbidities
The clinical complexity of these chronic conditions is further compounded by the fact that many have a wide array of overlapping symptoms and comorbidities. For instance, a subset of patients with ME/CFS, Long COVID, MCAS, POTS, and PTLDS frequently present with:
Overlapping symptoms include, but are not limited to:
- Brain fog
- Extreme fatigue
- Muscle/joint pain
- Sleep disturbances
- Psychiatric conditions such as:
- Mood disorders
- Anxiety disorders
- Major depressive disorder
Overlapping comorbidities include, but are not limited to:
- Fibromyalgia
- Autonomic dysfunction
- Mast cell activation syndrome
- Hypermobility spectrum disorders
- Hypermobile Ehlers-Danlos syndrome
The Hypermobility Comorbidity Overlaps
hEDS and hypermobility spectrum disorder (HSD) are connective tissue disorders that involve a spectrum of joint hypermobility, chronic pain (1), fatigue, and autonomic dysfunction.
Research shows that patients with joint hypermobility are at increased risk of also being diagnosed with one or multiple complex chronic conditions.
For instance, joint hypermobility appears in up to 30–57% of patients with ME/CFS (2-4), POTS (5, 6), Long COVID (7, 8), Fibromyalgia (9), and MCAS (10, 11) compared to 10–20% in the general population (12). In fact, the link between POTS, EDS, or HSD, and MCAS is so common that it is often referred to as the triad (or the “trifecta”).
Can Inflammation, Infection, and Mast Cell Activation Contribute to Connective Tissue Laxity?
Chronic inflammation is increasingly recognized for its role in connective tissue disorders like hEDS and HSD(11), but can inflammation from an infection or inappropriate mast cell activation increase or even cause hypermobility?
Emerging research in these conditions highlights immune dysregulation, elevated inflammatory markers (e.g., cytokines, chemokines, TGF-β), and complement pathway alterations in many patients (13). These contribute to extracellular matrix remodeling, tissue fragility, chronic pain, fatigue, and multisystem symptoms beyond pure structural defects.
How Inflammation Degrades the ECM and Connective Tissue
The extracellular matrix (ECM) is a scaffold of proteins, including collagen, fibronectin, laminin, and elastin. It provides structural support and mechanical stability for cells. Not only is the ECM found in every organ, but it’s also a major component of connective tissue.
When cells become inflamed due to a tissue injury or an infection, immune cells crawl through the tissue toward the affected site. Although these flexible immune cells can dynamically change shape and squeeze through the tightest of spaces, many ECM scaffolds are too dense for immune cells to penetrate.
Enzymes called proteases are released by the immune cells. These proteases act as molecular “pruning shears” that temporarily break down the ECM, thinning it without completely destroying it.
The proteases are released from white blood cells already present in the tissue (such as mast cells or macrophages) or from first responder cells that enter the tissue from the blood, such as activated neutrophils.
Inflammation does the following to the ECM:
- Upregulation of matrix metalloproteinases (MMPs): The metalloproteases are a family of enzymes that require a metal ion (hence the prefix “metallo”) to perform their function of cutting ECM components such as collagen and elastin (14).
- Mast cell activation (especially in MCAS): Activated mast cells release proteases (tryptase, chymase), histamine, and other mediators that further degrade ECM components.
- Oxidative stress: Reactive oxygen species (ROS) produced by ongoing inflammation can damage ECM proteins, worsening joint instability and pain (15).
While these processes allow immune cells to access damaged tissue for repair, persistent or dysregulated inflammation can lead to excessive ECM breakdown. One such form of immune system dysregulation is mast cell activation syndrome.
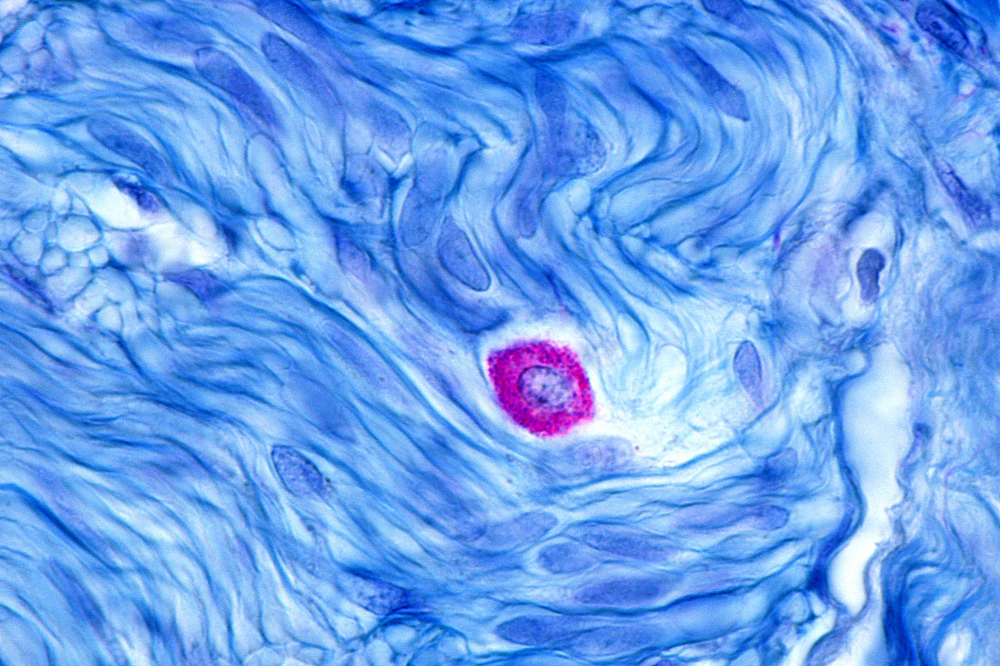
A microscopic image of a mast cell (stained purple) surrounded by muscle fibers (stained blue).
Mast Cell Activation Can Degrade Structural Components of Connective Tissue
Mast cells are ancient immune cells that evolved over 500 million years ago, predating all other immune cell types. They evolved before more specialized immune cells existed, forcing them to develop diverse mechanisms for immune protection against infections, injuries, and irritants.
Unlike less ancient specialized immune cell subsets, mast cells are considered the Swiss Army Knife of immune cells. With over 300 different receptors and the potential to release over 1200 different mediators, these immune cells play a role beyond allergic responses.
Unlike circulating immune cells, mature mast cells reside in the tissues, strategically positioned throughout virtually every organ and tissue, including connective tissue. This close proximity allows them to rapidly detect threats from infections, injuries, irritants, or neural signals within seconds.
When activated, mast cells degranulate, releasing a powerful “cocktail” of different mediators, including histamine, cytokines, prostaglandins, and crucially, proteases such as tryptase and chymase.
In a healthy state, this response is targeted and protective, helping contain threats and initiate repair. However, the accumulation of mutations in mast cell genes can result in excessive protease release, which can directly degrade collagen, elastin, fibronectin, and laminin, proteins that provide connective tissue with its strength (11).
For example, 28% of people with a mutation in alpha-tryptase also presented with hypermobility, a 2-fold increase over the background incidence in the general population (16). In addition, 46% of patients with the EDS, POTS, and MCAS triad had mutations associated with increased alpha-tryptase levels (16).
The Role of Alpha Tryptase in ECM Degradation
Alpha-tryptase is a protease released predominantly by mast cells that directly targets key components of the extracellular matrix (ECM) and connective tissue, including:
- Fibronectin: a glycoprotein that helps cells attach to the ECM.
- Type IV Collagen: a structural component in basement membranes.
- Proteoglycans: structures that help to hydrate and cushion joints.
Tryptase amplifies connective tissue degradation by activating MMPs that can further degrade the extracellular matrix of the connective tissues (including interstitial collagens, elastin, and laminin) (17). This is part of a larger cascade that creates a powerful amplification effect: tryptase not only degrades some targets directly but also triggers downstream enzymes that dismantle additional structural proteins.
The result is accelerated ECM remodeling and loss of tensile strength, which can significantly worsen joint hypermobility, instability, and related symptoms in susceptible individuals.
The Relationship Between Hereditary Alpha Tryptasemia (HαT) and POTS
Hereditary Alpha Tryptasemia (HαT) is a genetic anomaly causing chronically elevated alpha-tryptase levels. The persistent overactivity of this mast cell-derived tryptase is believed to drive excessive degradation of extracellular matrix (ECM) components and connective tissue remodeling (18).
In addition to causing increased connective tissue laxity, 34-46% of patients with HαT have autonomic symptoms and orthostatic intolerance(18).
Thus, too much of this mast cell-derived protease either contributes to POTS, exacerbates POTS symptoms, making it easier to diagnose, or excessive alpha-tryptase gene expression is epistatically influencing the expression of an unknown gene (or genes) that drives POTS progression (19).
Long COVID Increases Risk for Connective Tissue Disorders
Another clue that might explain the link between inflammation and hypermobility disorders is the association between acute inflammation from COVID-19 and hypermobility. Emerging evidence suggests that Long COVID, driven by persistent inflammation and immune dysregulation, can exacerbate or contribute to connective tissue complications, including worsening joint hypermobility (8).
For instance:
- Having generalized joint hypermobility or extreme hypermobility is a predictor of Long COVID (20).
- A case study revealed that five female patients with no prior hypermobility disorder diagnosis were later diagnosed with hEDS (or HSD) following a SARS-CoV-2 infection (21).
- A preprint study found that the rate of hEDS diagnosis was elevated following a SARS-CoV-2 infection, suggesting that the viral illness may exacerbate hypermobility symptoms (22).
- A large retrospective cohort study (n=354,527) linked COVID-19 to a substantially increased risk of autoimmune and autoinflammatory connective tissue disorders (23).
COVID-19 has also been shown to increase the expression of proteases that degrade components of connective tissue (24). However, this is not a unique feature of SARS-CoV-2, as most infections force immune cells to remodel connective tissue so they can crawl through the tissue to neutralize the pathogen or pathogen-infected cells.
Is the link between hypermobility and Long COVID caused by built-in pathogenic features of the SARS-CoV-2 virus, or is the acute inflammatory spike contributing to this relationship?
Therapies that relieve inflammation can reduce symptom severity in a subset of patients
If a subset of joint hypermobility disorders is attributed to inflammation, then treating the inflammation would improve the hypermobile symptoms.
Peer-reviewed evidence shows symptom relief from anti-inflammatory interventions, supporting inflammation’s role:
- MCAS stabilizers, such as cromolyn sodium, ketotifen, and antihistamines, provide the strongest support, reducing mediator release and improving flushing, tachycardia, orthostatic symptoms, and hypermobility-related pain in 60-80% of responders in MCAS-overlap POTS/HSD cases (11, 25).
- One case report determined that IVIG (intravenous immunoglobulin therapy targeting inflammation) in addition to naltrexone and antibiotics successfully treated a patient with POTS and MCAS (26), while a case series of six patients with POTS given one IVIG infusion per month for six months resulted in a reduction of symptom severity by 40% (27).
These responses are strongest in inflammatory subsets, suggesting that chronic inflammation can worsen tissue degradation and multisystem symptoms in a subset of people with hEDS/HSD.
However, larger randomized controlled trials are needed to rigorously assess whether attenuating the chronic inflammation can improve symptoms of hEDS or HSD.
The Complexity of Complex Chronic Conditions
These medical conditions are characterized as complex for a reason. They often involve different biological drivers that cause a set of overlapping symptoms and comorbidities, leading to overlapping (different) treatment responses.
This means that identical symptoms and comorbidities from two different POTS patients might be attributed to different pathological mechanisms of disease. For example, the presence of autoantibody responses (28), pregnancy (29), or excessive ECM breakdown (16).
Not only are the drivers of disease, symptoms, and comorbidities different from person to person, but the high degree of diversity in disease severity makes them difficult to study.
The most severely affected patients are often excluded from clinical studies because of inclusion and exclusion criteria. This results in outcomes that are not representative of the entire patient population, thereby leading to an incomplete understanding of the disease spectrum.
There has yet to be a single study demonstrating a unifying disease mechanism or mechanisms to explain the diagnostic overlap of hEDS, POTS, MCAS, and Long COVID. While chronic inflammation may be a common thread in a subset of patients, it’s important to recognize the roles of other pathological features of the disease, such as brainstem compression, mitochondrial dysfunction, oxidative stress, and HPA axis disruptions. We argue that we need a different approach to studying these complex diseases.
A Collaborative Approach To Studying Complex Chronic Diseases
We must embrace the diversity of these complex diseases rather than forcing patients into rigid diagnostic silos. Instead of labeling only those who perfectly match a textbook definition and dismissing the rest as outliers, we should recognize that many patients present with meaningful variations of the same underlying condition.
Accepting this diversity allows for more inclusive, accurate, and compassionate care that reflects the real-world spectrum of illness.
We at the Brain Inflammation Collaborative are taking a collective approach toward viewing these complex chronic conditions as disease clusters. A key part of this approach involves our digital health platform, the unhide® Solve Together Unified Platform, designed to uncover hidden connections between seemingly unrelated symptoms and diagnoses, and to advance patient-centered research that drives better diagnosis and treatment.

unhide® is a patient-centered research platform designed to help individuals track symptoms, identify patterns, and contribute to meaningful medical research—particularly for those living with complex, often misunderstood conditions like PANS and PANDAS. By combining real-time symptom tracking with community-driven data, unhide empowers patients and caregivers to transform their lived experiences into insights that inform peer-reviewed studies and improve future care. The platform bridges the gap between patients and researchers, ensuring that what families notice and report directly shapes the science meant to support them. Find out more at unhidenow.org.
FAQ
Can you have both ME/CFS and Ehlers-Danlos syndrome?
Yes, you can have both ME/CFS (myalgic encephalomyelitis/chronic fatigue syndrome) and Ehlers-Danlos syndrome (EDS), particularly the hypermobile type EDS (hEDS). Studies and clinical observations show significant overlap, with joint hypermobility appearing in up to 50% of ME/CFS patients, far higher than the 10–20% in the general population (2-4). This comorbidity often involves shared features like chronic fatigue, autonomic dysfunction (e.g., POTS), and connective tissue-related pain, though the exact relationship is still under study.
What is neuroinflammation, and what are the symptoms?
Neuroinflammation is an inflammatory response within the brain or spinal cord (central nervous system). It serves as a protective mechanism against injury, infection, or toxins, but can become harmful when chronic or excessive, contributing to neuronal damage, neurodegeneration, or persistent symptoms in conditions like ME/CFS, Long COVID, and others. Common symptoms include:
- Brain fog (difficulty concentrating, memory issues, slowed thinking)
- Sometimes numbness, tingling, weakness, or balance problems
- Mood changes (anxiety, depression, irritability)
- Cognitive impairment (poor focus, confusion)
- Fatigue and low energy
- Sleep disturbances
- Headaches
In chronic illnesses, these often overlap with “sickness behavior” described as profound tiredness and post-exertional malaise.
How is Long COVID related to ME/CFS?
Individuals with Long COVID present with a collection of symptoms that mirror ME/CFS, such as persistent fatigue, post-exertional malaise (PEM), unrefreshing sleep, and cognitive impairment (“brain fog”). Both conditions often follow a viral trigger (SARS-CoV-2 for Long COVID; various infections such as EBV or HHV6 for ME/CFS) and share proposed mechanisms, including chronic inflammation, immune dysregulation, neuroinflammation, autonomic dysfunction (e.g., POTS), mitochondrial dysfunction, and potential brainstem dysfunction (30, 31).
Can inflammation cause hypermobility?
Chronic or dysregulated inflammation caused by infections (e.g., post-viral, such as Long COVID), mast cell activation (MCAS), or immune dysregulation can increase the abundance of proteases that degrade the structural components of the extracellular matrix (ECM). Persistent ECM breakdown may lead to increased connective tissue laxity, joint hypermobility, instability, and related symptoms, such as pain and autonomic issues, in a subset of people (8, 18, 26).
Is there treatment for neuroinflammation?
There is no universal treatment for neuroinflammation. However, neuroinflammation can be successfully treated if the underlying cause is properly identified and addressed. For example, treating PANDAS/PANS neuroinflammation (post-infectious autoimmune neuropsychiatric disorders) often involves IVIG, antibiotics, or corticosteroids to target bacterial or viral triggers and the dysregulated immune response, while autoimmune encephalitis (AE) typically uses IVIG, high-dose corticosteroids, rituximab, or plasmapheresis to target autoantibody-mediated brain inflammation (32). In ME/CFS-related neuroinflammation, management focuses on addressing potential brainstem compression or intracranial pressure (33), though the evidence is emerging and not standardized.
- B. Kumar, P. Lenert, Joint Hypermobility Syndrome: Recognizing a Commonly Overlooked Cause of Chronic Pain. Am J Med 130, 640-647 (2017).
- K. Mudie, A. Ramiller, S. Whittaker, L. E. Phillips, Do people with ME/CFS and joint hypermobility represent a disease subgroup? An analysis using registry data. Frontiers in Neurology Volume 15 – 2024, (2024).
- L. M. van Campen, P. C. Rowe, F. C. Visser, The myalgic encephalomyelitis/chronic fatigue syndrome patients with joint hypermobility show larger cerebral blood flow reductions during orthostatic stress testing than patients without hypermobility: A case control study. Medical Research Archives 9, (2021).
- B. Bragée et al., Signs of Intracranial Hypertension, Hypermobility, and Craniocervical Obstructions in Patients With Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Front Neurol 11, 828 (2020).
- J. R. Boris, T. Bernadzikowski, Prevalence of joint hypermobility syndromes in pediatric postural orthostatic tachycardia syndrome. Autonomic Neuroscience 231, 102770 (2021).
- E. Grigoriou, J. R. Boris, J. P. Dormans, Postural orthostatic tachycardia syndrome (POTS): association with Ehlers-Danlos syndrome and orthopaedic considerations. Clin Orthop Relat Res 473, 722-728 (2015).
- C. D. Eccles JA, Quadt L, Hakim AJ, Gall N, Bowyer V, in Case-control evidence from the British COVID Symptom Study Biobank. . (BMJ Public Health, 2024).
- R. Ganesh, B. Munipalli, Long COVID and hypermobility spectrum disorders have shared pathophysiology. Frontiers in Neurology Volume 15 – 2024, (2024).
- M. Acasuso-Díaz, E. Collantes-Estévez, Joint hypermobility in patients with fibromyalgia syndrome. Arthritis Care Res 11, 39-42 (1998).
- L. B. Afrin et al., Diagnosis of mast cell activation syndrome: a global “consensus-2”. Diagnosis (Berl) 8, 137-152 (2021).
- A. Monaco, D. Choi, S. Uzun, A. Maitland, B. Riley, Association of mast-cell-related conditions with hypermobile syndromes: a review of the literature. Immunol Res 70, 419-431 (2022).
- M. Castori et al., A framework for the classification of joint hypermobility and related conditions. Am J Med Genet C Semin Med Genet 175, 148-157 (2017).
- M. Griggs et al., Proteomic discoveries in hypermobile Ehlers–Danlos syndrome reveal insights into disease pathophysiology. ImmunoHorizons 9, (2025).
- H. S. Lee, W. J. Kim, The Role of Matrix Metalloproteinase in Inflammation with a Focus on Infectious Diseases. Int J Mol Sci 23, (2022).
- J. A. Eble, F. F. de Rezende, Redox-relevant aspects of the extracellular matrix and its cellular contacts via integrins. Antioxid Redox Signal 20, 1977-1993 (2014).
- J. J. Lyons et al., Elevated basal serum tryptase identifies a multisystem disorder associated with increased TPSAB1 copy number. Nat Genet 48, 1564-1569 (2016).
- V. Payne, P. C. Kam, Mast cell tryptase: a review of its physiology and clinical significance. Anaesthesia 59, 695-703 (2004).
- K. T. Luskin, A. A. White, J. J. Lyons, The Genetic Basis and Clinical Impact of Hereditary Alpha-Tryptasemia. J Allergy Clin Immunol Pract 9, 2235-2242 (2021).
- J. Huang et al., Hereditary alpha-tryptasemia in patients with postural orthostatic tachycardia syndrome. J Allergy Clin Immunol Pract 12, 528-529.e521 (2024).
- R. A. Torok, J. Lubell, R. M. Rudy, J. Eccles, L. Quadt, Variant connective tissue as a risk factor for long COVID: a case-control study of data from a retrospective online survey of adults in the USA and UK. BMJ Public Health 3, e002949 (2025).
- M. Yang et al., Long COVID and the Diagnosis of Underlying Hypermobile Ehlers-Danlos Syndrome and Hypermobility Spectrum Disorders (P5-4.014). Neurology 102, 2478 (2024).
- M. L. Pearson et al., Understanding Comorbidities in Hypermobile Ehlers-Danlos Syndrome: Could a Viral Infection Lead to a Diagnosis? medRxiv, 2025.2010.2022.25338573 (2025).
- Y.-W. Heo, J. J. Jeon, M. C. Ha, Y. H. Kim, S. Lee, Long-Term Risk of Autoimmune and Autoinflammatory Connective Tissue Disorders Following COVID-19. JAMA Dermatology 160, 1278-1287 (2024).
- S. L. Murphy et al., Extracellular matrix remodelling pathway in peripheral blood mononuclear cells from severe COVID-19 patients: an explorative study. Front Immunol 15, 1379570 (2024).
- L. Yao et al., Association of postural orthostatic tachycardia syndrome, hypermobility spectrum disorders, and mast cell activation syndrome in young patients; prevalence, overlap and response to therapy depends on the definition. Front Neurol 16, 1513199 (2025).
- L. B. Weinstock, J. B. Brook, T. L. Myers, B. Goodman, Successful treatment of postural orthostatic tachycardia and mast cell activation syndromes using naltrexone, immunoglobulin and antibiotic treatment. BMJ Case Reports 2018, bcr-2017-221405 (2018).
- B. Rodriguez, R. Hoepner, A. Salmen, N. Kamber, W. J. Z’Graggen, Immunomodulatory treatment in postural tachycardia syndrome: A case series. European Journal of Neurology 28, 1692-1697 (2021).
- H. Li et al., Autoimmune basis for postural tachycardia syndrome. J Am Heart Assoc 3, e000755 (2014).
- K. M. Bourne et al., Symptoms of postural orthostatic tachycardia syndrome in pregnancy: a cross-sectional, community-based survey. Bjog 130, 1120-1127 (2023).
- A. D. Proal, M. B. VanElzakker, Long COVID or Post-acute Sequelae of COVID-19 (PASC): An Overview of Biological Factors That May Contribute to Persistent Symptoms. Front Microbiol 12, 698169 (2021).
- J. D. Wood et al., Brainstem dysfunction as a potential etiology of ME/CFS and long COVID: A mechanical basis. Medical Hypotheses 206, 111812 (2026).
- E. Lancaster, The Diagnosis and Treatment of Autoimmune Encephalitis. J Clin Neurol 12, 1-13 (2016).
- T. Nelson, L. X. Zhang, H. Guo, L. Nacul, X. Song, Brainstem Abnormalities in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: A Scoping Review and Evaluation of Magnetic Resonance Imaging Findings. Front Neurol 12, 769511 (2021).


No Comments